Welding Of Aluminium
Description
This section is from the book "Welding And Cutting Metals By Aid Of Gases Or Electricity", by L. A. Groth. Also available from Amazon: Welding and cutting metals by aid of gases or electricity.
Welding Of Aluminium
The welding of aluminium has met with great difficulties by reason of the great ability of oxidation of the material. The moment it has been prepared and cleansed, the aluminium is at once covered with aluminium oxide, which prevents the pieces fusing together. By using a flux, however, the oxidation skin is dissolved, and a dross is simultaneously formed which enables the metal to flow and make a perfect weld. The weld remains indifferent in solutions of common salt and soda, and is in every respect comparable with rolled aluminium.
There are various systems for soldering aluminium, and for welding; for instance, those of M. D. Schocp and W. C. Heraeus. The former is described as follows: -
The numerous alloys of the aluminium-copper-tin or tin-bismuth-copper type, which have been used for soldering aluminium at moderately low temperature, are all open to the same objection, that the soldered joint slowly loses its cohesion, i.e., mechanical strength. This is due to the fact that aluminium, particularly in the presence of water, compares unfavourably with other metals on account of the "electrolytic local action," the aluminium becoming slowly decomposed.

Fig. 16.
Apart from the admixture of the deoxydising substance, which plays the same part as borax does in soldering, the welding of aluminium is similar to lead-burning. The welding requires no special preparations nor any plant beyond the equipment of tools necessary for the efficient handling of the aluminium.
The difficulty to be overcome was, therefore, to find means whereby welding of aluminium could be made without the addition of another metal, eliminating thereby electrolytic difficulties, and ensuring the same physical and chemical properties of the weld as those of the bar or plate to be welded.
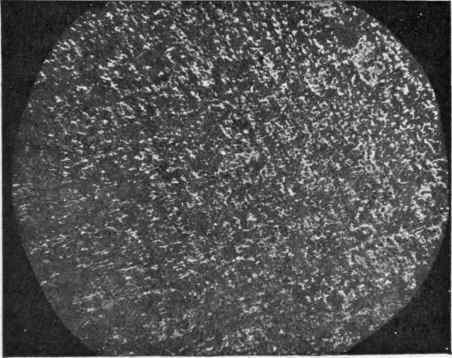
The welding of aluminium has been successfully accomplished by the Schoop's process. The Government Institution, Le Laboratoire du Conservatoire National des Arts et Metiers a Paris, has made some microphotographic tests, reproductions of which are given in Fig. 17 of the autogenously welded aluminium, and Fig. 18 of the unwelded aluminium.
Again, it is well known that aluminium, like lead, is attacked by the atmosphere, and becomes coated with a layer of oxide, but whereas in the case of lead this oxidation-product can easily be removed, the coating on aluminium has hitherto resisted all practicable modes of removal. By means of a flux, however, the very tough layer of oxide is immediately removed under the blowpipe flame, and the surfaces thus cleaned and fused are readily united and produce a direct union of aluminium with aluminium without in any way impairing the original metal.
The principal point is that the blowpipe flame be of sufficiently high temperature to fuse the surface of the aluminium. For aluminium sheeting 1 mm. thick coal-gas and oxygen would suffice, whilst for stout aluminium sheeting acetylene and oxygen would be preferable.
Fig. 17.

Fig. 18.
Constants of Aluminium,
Atomic weight.... 27'0
Specific gravity (cast) . . . . . . 2.6 at 4 per cent.
,, „ (rolled or hammered) . . . 2.67 ,,
Melting point.... 650° C.
Tensile and compressive strength of cast aluminium . 10 at 12 kilos per sq. mm.
,, ,, ,, rolled ., . 25 at 27 kilos per sq. mm.
Electric conductivity (copper = 100) . . . . 60
Continue to:


