Chapter IV. Blowpipes
Description
This section is from the book "Welding And Cutting Metals By Aid Of Gases Or Electricity", by L. A. Groth. Also available from Amazon: Welding and cutting metals by aid of gases or electricity.
Chapter IV. Blowpipes
General Remarks - Daniel's Burner - Hydrogen Blowpipe - Schuckert - L'Oxbydrique Internationale - Oxy-hydrogen Blowpipe Plant - Draeger-Wiss - Acetylene Blowpipe - High-pressure Type - Low-pressure Type - Acetylene Illuminating Company, Limited - Fouche Blowpipe - Oxy-acetyleue Blowpipe Plant.
General Remarks
The blowpipe is an instrument by means of which the operator approaches the metal upon which the work is intended to be done, while by the old system the metal had to be brought to the operator.
The blowpipe provides thus a method of dealing very simply with an immense number of metallurgical operations, which have until quite recently been carried out under less favourable conditions.
The various systems of welding seem, however, to require a special form of blowpipe ; at least, each system claims its own construction as a speciality. Blowpipes may therefore be classed according to the combustible gas which they employ, such as hydrogen, acetylene, ordinary coal gas, and naphtha ; but all of these utilise the same supporter of combustion gas, oxygen.
The facility by which oxygen and hydrogen can be obtained for commercial purposes drew attention to the possibilities of the oxy-hydrogen blowpipe as a welding agent. At first
Daniel's burner was used, the gases being mixed at the mouth of the burner before combustion, but without satisfactory results. Then it was remembered that if the mixing of the gases took place before the egression from the burner, their combustion would produce a higher temperature. This is the principle upon which all blowpipes are now being constructed. So far as the oxy-hydrogen system is concerned, the mixing of the two gases may take place either outside or inside the blowpipe. Schuckert, for instance, mixes the gases before they enter the blowpipe, and employs for this purpose a special mixing chamber, placed about 1 metre from the blowpipe. The oxygen and hydrogen are passed in proper proportions through rubber tubes into the mixing chamber. The mixture travels from there, by means of a rubber tube, into the blowpipe, ready for combustion.
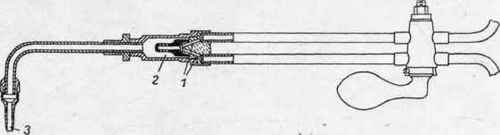
Fig. 83.
In most cases, however, the gases are mixed in the blowpipes. This may be considered to be the safest way to obtain a proper mixture and the regulation of the gases. The Societe Oxhydrique Internationale mix the oxygen and hydrogen in a chamber formed in the mixing part of the tube, which also carries the burner. Drager-Wiss, according to the journal Vulcan, modifies the construction by placing the channels 1 and 2 (Fig. 83) for the oxygen and hydrogen in an oblique position towards one another, and by producing a suction, prevents the gases to pass into the tubes of one another ; the mixture of the two gases takes place in a larger chamber before it enters the nozzle 3 for combustion.
The Draeger-Wiss blowpipe is based upon the injector

No. 1, for 1 mm.

No. 2, for 2 mm.
No. 3, for 3 mm.

No. 5, for 4 - 5 mm.

No. 7, for 5 - 7 mm.

No. 10, for.
8 - 10 mm.

No. 13, for 10 - 13 mm.

No. 16, for.
13 - 16 mm.

No. 20, for 16 - 20 mm.

No. 25, for 20 - 25 mm.
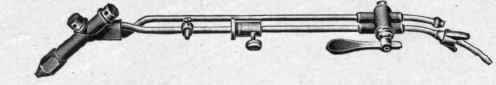
Fig. 84. - Acetylene Blowpipe, Draeger-Wiss, Model 1908.
principle, and although very simple in its construction it nevertheless embodies the qualities required of an effective blowpipe. Fig. 84 shows the various sizes of the model of 1908 applicable for different thicknesses of metal. The blowpipe is as suitable for hydrogen as for acetylene, and is very extensively used abroad.
In case the mixture is too poor in oxygen, the combustion becomes very slow. This can easily be verified by filling a glass tube with a gas mixture poor in oxygen. By lighting the mixture at one end of the tube the flame may be seen travelling towards the other end of the tube. The more oxygen added to the mixture, the faster and more complete will the combustion be; when it has reached its maximum, i.e., hydrogen and oxygen in the proportion of 2 : 1, the activity amounts to about 2,800 metres per second, resulting in an explosion, but without dangerous effects. Oxy-hydrogen blowpipe plant is illustrated in Fig. 82 on page 141.
When the gases are burned in the proportion of two volumes of hydrogen to one volume of oxygen, the proportions required for complete combustion, the temperature of the flame produced is about 6.000° Fahr. In order, however, to ensure a non-oxidising flame which cannot injuriously affect the character of the metal operated upon, it is found that the gases must be burned in the proportion of about four volumes of hydrogen to one volume of oxygen, so that the temperature of the flame produced by this blowpipe in actual operation is probably about 4,000° Fahr.
The oxy-hydrogen blowpipe thus constructed supplied to a great extent what was wanted, but it has, like all other constructions of blowpipes, some drawbacks, which, to a small extent, will reduce its general use, the chief one being that of not producing, for certain purposes, a sufficiently high temperature ; besides, an error in the mixture, however small, will change the nature of the flame into an oxydising, or reducing one, consequent upon an excess or shortage of oxygen. Nevertheless, the oxy-hydrogen blowpipe renders quite as great service and offers similar advantages as those of other blowpipe systems, which is testified by the extreme extent to which it is being used on the Continent.
Continue to:


