Oxygen. Part 2
Description
This section is from the book "Welding And Cutting Metals By Aid Of Gases Or Electricity", by L. A. Groth. Also available from Amazon: Welding and cutting metals by aid of gases or electricity.
Oxygen. Part 2
Professor Garuti's Process having many advantages is therefore described here: -
The principal points to consider are:
1. Reduction to a minimum of the electro-motive force required, and
2. Perfect separation of gases evolved.
In order to realise the first condition it is necessary to determine the most favourable composition of the electrolyte, that is, the liquid to be decomposed, and the proper separation of the electrodes. In Fig. 1 the electrodes are shown to be separated by the porous diaphragm D, but, as the circulation of the electrolyte is essential for the continuity of the process, it should be so constructed as to permit this; in order to assure the separation of the gases, they should not be permitted to pass through it, but to reduce the resistance the diaphragm should be a conductor.
The principal conditions required of a perfect diaphragm is that it should be permeable for water, impenetrable for gases, and a good conductor of electricity. It is evident that the construction of the diaphragm is the main difficulty. Various materials for the same have been employed, such as biscuit-baked porcelain, pipe clay, plates of carbon, amianthus, but they have all offered disadvantages, particularly in respect of the electrical resistance and the inability of preventing the gases to pass through.
Garuti has suggested the application of metallic diaphragms, which also have been found to give satisfactory results. If a metal plate is placed between the two electrodes (Fig. 2) it is influenced by the current, the positive pole formed opposite the negative one in each compartment, whereby hydrogen and oxygen will be generated, i.e., explosive gas. Garuti has discovered, however, that this will not take place if the electromotive force does not exceed three volts and the density of the current remains under two amperes per square decimetre of electrode; under such circumstances the diaphragm remains passive, and by reason of its low resistance it is possible to work with a potential under three volts.
It remains then to assure the circulation of the electrolyte. On a first examination it is found that the better the circulation is assured the less is the resistance. It seemed sufficient to leave a space between the diaphragm and the bottom of the tank, and it has been found that the lower edge of the diaphragm should not reach below those of the electrodes (Fig. 3). A mixture of gas, however, takes place, forming upon the electrodes a certain volume of bubbles, which, increasing and disengaging themselves, pass direct to the surface. Other bubbles, very small, almost microscopical, detach themselves, as soon as they are formed, from the electrodes and remain in suspension, probably by reason of their extreme tenuity, and darken or cloud the electrolyte, descend, and pass under the electrodes, turning into the neighbouring compartment, from whence the mixture of gas. In order to prevent this mixing it is found sufficient simply to lower the diaphragm until the proper resistance has been obtained.
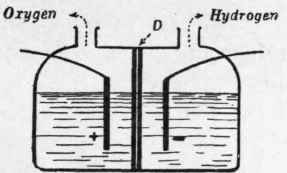
Fig. 1.
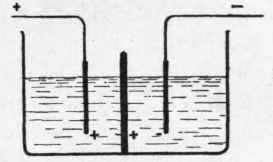
Fig. 2.
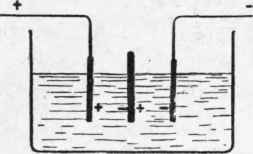
Fig. 3.
In order to assure the circulation of the electrolyte the metallic diaphragm is perforated. Experience has proved that these perforations can have a diameter up to one m.m. each, and that they should be as numerous as possible and be united by means of a band some centimetres high, and placed in front of the electrode. Strangely enough, these perforations, being large enough to circulate the electrolyte, are almost impermeable for the gases, probably by some capillary reason. The metallic diaphragms, by being welded together, form cells (Fig. 4), each containing an electrode, and fulfil thereby every condition that is required.
By an ingenious arrangement the number of weldings is reduced to a minimum. The cells of the apparatus are placed side by side, with their lower ends open entirely, but their upper parts open about half their length. All the cells containing an anode are half open to the left side, and those containing a cathode are half open to the right. A bell or funnel enclosing the left ones collects oxygen, and a similar funnel containing the right cells collects hydrogen (Fig. 5). In apparatus of certain sizes the diaphragms as well as the electrodes are rigidly kept in their places by wooden combs placed at their lower ends.
In the construction of the apparatus lead is used, in some special cases, with an electrolyte composed of water and an acid, but in general iron or steel is used, and then with an electrolyte composed of a solution of soda or caustic potash ; the latter presents a smaller resistance, but is more expensive. The solution has a minimum resistance of 15 per cent. for soda, but generally 25 per cent., and 29 per cent. for potash.
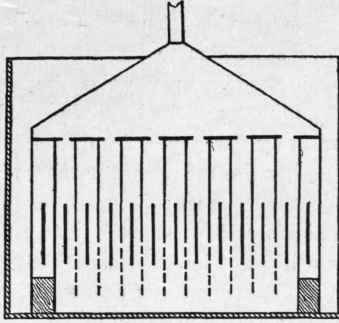
Fig. 4.
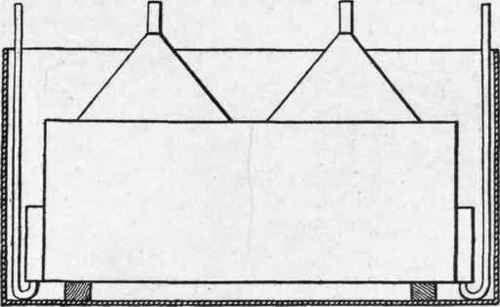
Fig. 5.
In order to facilitate the liberation of the gas bubbles and to avoid their passing through the perforated diaphragms the solution should be very concentrated.
The iron is not quite indifferent to the action of the alkali, and Professor Eric Gerard has found the wear and tear of the anodes in Garutis apparatus to amount to 15 per cent. of the weight of the anode. In practice the anodes have a thickness of 0.7 millimetre at the beginning, and require to be exchanged after every three years' working; consequently a very small loss; otherwise the apparatus offers no alteration, the electrolyte does not change; should it, however, in length of time become highly carboniferous, it is easy to generate the solution by some lime. The anodes as well as the wooden combs require to be exchanged every three years; the costs of maintenance are therefore practically nil.
Continue to:


